|
Bib. Me: Free Bibliography & Citation Maker. Select style& search. Select style & search. Search for a book, article, website, film, or enter the information yourself. How Appetite Effects Your Weight Cellular energy balance sensor AMPK AMPK is a key sensor and regulator of energy balance at the cellular level. Its activity is regulated by the ratio of AMP: ATP inside a cell.  
Documentation and reference library for ZURB Foundation. JavaScript, CSS, components, grid and more. Research on major depression has confirmed that it is caused by an array of biopsychosocial and lifestyle factors. Diet, exercise and sleep are three such influences. Gluten-brain connection. The Celiac Disease of Mental Illness (derived in large part from a lecture James V. Croxton, M.A. For the purpose of this article, research on meditation concerns research into the psychological and physiological effects of meditation using the scientific method. 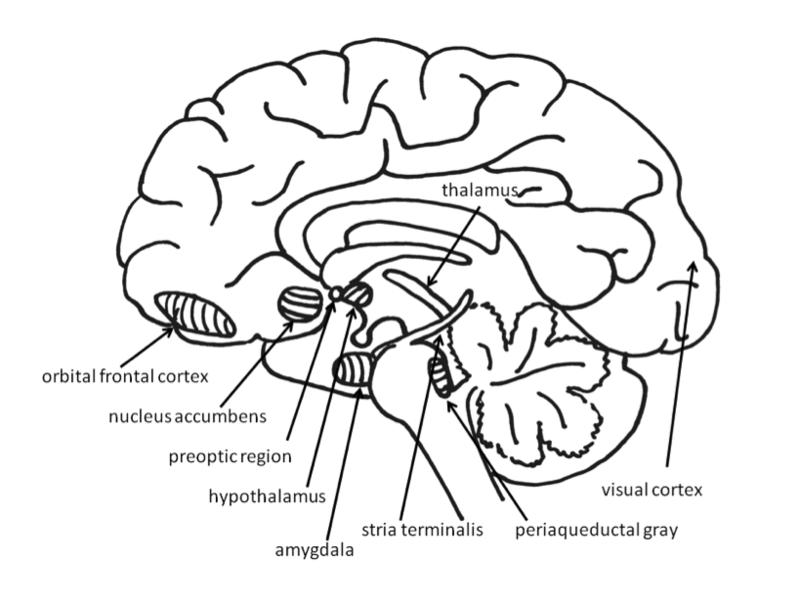  
A higher AMP: ATP ratio indicates a lower energy supply and vice versa. AMPK is a heterotrimer comprised of a catalytic alpha- subunit and regulatory beta- and gamma- subunits. The gamma- subunit can bind to AMP at a higher AMP: ATP ratio or to ATP at a lower AMP: ATP ratio. Binding to AMP causes the phosphorylation of threonine (Thr- 1.   
LKB1, Ca. MKK beta or TAK1, and leads to the activation of AMPK. Once activated, AMPK turns on catabolic pathways that generate ATP and turns off anabolic pathways that consume ATP (Fig. AMPK signaling in energy balance regulation. Green arrows indicate AMPK activation induced by negative energy balance while red arrows indicate AMPK inhibition by positive energy balance. Abbreviations: LKB1, serine threonine kinase 1. Ca. MKK . During times of energy shortage, AMPK is activated in peripheral tissues to favor local ATP production, and in the brain, to stimulate neuroendocrine pathways that increase food intake. Thus AMPK activation prepares the whole organism for energy acquisition (Deshaies, 2. There is no doubt that mindfulness meditation has many benefits, but what's the scientific evidence behind how it affects our bodies? You can read some of the latest. 1 Laboratory of Molecular and Cellular Neuroscience, The Rockefeller University, New York, NY, USA 2 Department of Physiology and Biophysics, School of Medicine and. Agonists of AMPK have been under development by pharmaceutical companies as potential treatments for obesity (Hardie, 2. They are classified as either episodic or tonic signals. Episodic signals are short- term and produced between meals (eating episodes). 
They impact our decisions about when and how much to eat. The major episodic signals described below include the “hunger signal,” ghrelin, and the “appetite suppressors,” CCK, GLP- 1, PYY and amylin. Tonic signals are generated by the body's response to nutrition status in the long term (days or weeks). They potentiate the magnitude of episodic signals and cause us to subconsciously adjust meal frequency and size (Fig. Variations of the genes involved in these signaling processes, such as LEP and LEPR, are normally rare but are associated with severe obesity symptoms when they occur. In the human brain, information must be stored in order for learning to take place. So, the areas of the brain that are most important to.The main energy balance control signals in human body (adopted from Blundell et al, 2. Ghrelin is a peptide hormone primarily produced by a group of special cells (called the P/D1 cells) in the upper section of the stomach. Its expression is inhibited by high glucose concentration in the P/D1 cells. When blood glucose levels drop below a threshold (comparable to hypoglycemia level, about 1m. M), the expression of ghrelin is activated. It is released into the bloodstream and eventually reaches the arcuate nucleus of the hypothalamus (ARC) where it activates the expression of agouti- related protein (Ag. RP) and neuropeptide Y (NPY). Both Ag. RP and NPY are signaling molecules that stimulate the orexigenic neuron, generating the “hunger signal.” Besides glucose, ghrelin release is also regulated by a number of other signals. For example, hormones such as epinephrine and norepinephrine stimulate ghrelin release while insulin and somatostatin (a peptide hormone produced in the hypothalamus) inhibit release (Fig 3). In addition to the “hunger signal,” ghrelin also plays an important role in growth hormone secretion, cognitive adaptation, learning, and blood vessel relaxation. Ghrelin is encoded by the GHRL gene. The GHRL gene product is a 1. PC) 1/3. Obestatin is involved in satiety and decreased food intake in rats (Zhang et al, 2. The 2. 8 amino acid ghrelin peptide is inactive until it is acetylated on Ser. C8–C1. 0) fatty acid. The acylation is catalyzed by the enzyme ghrelin O acyltransferase (GOAT) and is required for ghrelin binding to the ghrelin receptor (GRLN R). Therefore, the bioavailability of ghrelin is determined by the activities of PC 1/3 as well as that of GOAT. It is believed that the acetylation and secretion of ghrelin are regulated differently. Several mutations and polymorphisms of the GHRL gene have been associated with various degrees of obesity, but reports in literature are inconclusive and inconsistent. However, increased ghrelin levels have been reported in individuals with anorexia, which suggests ghrelin resistance may play a role in this condition. More than a dozen drugs targeting ghrelin, GRLN R, or GOAT are being developed for appetite control or growth regulation (Casta. Ghrelin regulation by the neuronal hormones, glucose and insulin (adopted from Dong & Brubaker et al, 2. Cholecystokinin (CCK) is an incretin. Incretin is a group of gastrointestinal hormones that increase insulin in response to intestinal nutrients. CCK is released in the small intestine where it acts in the vagal nervous system to increase satiety. Fatty acids from digested fat stimulate CCK through the G- protein- coupled receptor GPR4. The release of CCK activates CCK receptors on the vagal neuron in the stomach. The signal is then transmitted from the vagus nerve to the brain stem where it is relayed to the hypothalamic region and is integrated with other signals to determine whether to stop or continue eating. Several drugs target the CCK pathway for weight control. Synthetic CCK analogues or CCK- A receptor antagonists (e. Glucagon- like peptide- 1 (GLP- 1) is another incretin hormone. It is synthesized in the gut and released into the bloodstream. In addition to enhancing glucose- dependent insulin biosynthesis and insulin secretion through the GLP- 1 receptor in the pancreatic . GLP- 1 also delays gastric emptying (transfer of the partially digested food mixture from the stomach to the small intestine) and prolongs the feeling of fullness. GLP- 1, GLP- 2 and oxyntomodulin are three hormones cleaved from the same protein proglucagon by prohormone convertase PC1/3, and are co- secreted by the same L cell. The effect of GLP 2 is localized in the gastrointestinal tract. Its main functions include increasing small and large intestinal weight, crypt- villus height and mucosal surface area, and nutrient absorption. Oxyntomodulin is believed to have the same function as GLP- 1, acting via the same receptors. Carbohydrates and fats are the main nutrients that stimulate the release of these hormones. Other signals, such as neurotransmitter acetylcholine and hormones insulin and leptin, also regulate their production (Fig. Production and regulation of GLP- 1, GLP- 2, oxyntomodulin and PYY by food nutrients and hormones (adopted from Dong & Brubaker et al, 2. PYY (Peptide tyrosine tyrosine) 1- 3. The bioactive peptide PYY 3- 3. PYY 1- 3. 6 by the enzyme dipeptidyl peptidase IV (DPP- 4). PYY3- 3. 6 is released from the same L- cells alone with GLP- 1 after nutrient stimulation (Fig. Through the vagal nerve neurons, it delays gastric emptying and stimulates the anorexigenic neurons in the hypothalamus to suppress appetite. At their highest physiological concentration (after a meal), PYY3- 3. Batterham et al 2. The natural plasma levels of PYY are generally lower in obese people. Pharmaceutical companies have developed synthetic PYY 3- 3. Amylin is a 3. 7 amino acid pancreatic hormone that helps reduce food intake through the medulla of the brainstem and by delaying gastric emptying. When blood glucose levels are high, amylin is released from the pancreatic beta cells along with insulin. It is believed to function synergistically with insulin to control blood glucose levels. A human amylin analogue, pramlintide reduces appetite in both lean and obese people (Harrold et al, 2. Leptin is a 1. 6 Kd hormone produced by fat tissue. It is a principal long- term regulator of energy balance by functioning on both food intake (satiety) and body fat metabolism. Encoded by the LEP gene, leptin is secreted into the bloodstream and delivered to the leptin receptor at the hypothalamus, medulla and other sites. The binding of leptin to its receptor in the hypothalamus activates the JAK/STAT signal transduction pathway. Ultimately the expression of genes involved in appetite inhibition and fat burning (such as POMC, GLP- 1 and AMPK) are turned on and the expression of genes involved in appetite stimulation (such as Ag. RP and NPY) are turned off. In general, leptin levels in the bloodstream are proportional to body fat, and the amount of leptin entering the CNS is proportional to its plasma concentrations. Therefore, increased body fat will lead the body to decrease food intake and increase energy expenditure by increasing energy metabolism. However, like insulin resistance, leptin resistance is becoming an epidemic in overweight or obese populations. Currently, the mechanism of leptin resistance is not well understood. Mutations in LEP or LEPR genes are responsible for rare but severe obesity symptoms. The homeostatic system is located in the hypothalamus and the brain stem while the hedonic system is distributed throughout the limbic regions including the cortex and prefrontal cortex (Fig. Several sections of the hypothalamus are at the center of homeostatic regulation (Fig. B). The ARC (arcuate nucleus) contains two populations of neurons. The first population co- expresses the peptides NPY (neuropeptide Y) and Ag. RP (agouti- related peptide) while the second contains POMC (pro- opiomelanocortin) and CART (cocaine- and amphetamine- regulated transcript). NPY and Ag. RP increase appetite by stimulating orexigenic neurons while . Balanced orexigenic and anorexigenic neuron activity ensures the homeostasis of energy balance. The hedonic system controls not only food preferences but also many other emotional and cognitive aspects in relation to happiness. Together, these two systems make the final decision when it comes to what, when and how much to eat. Many obesity risk genes discovered in the last decade, such as those shown in Table 2, are involved in CNS signaling and appetite regulation. The appetite control system in the brain. In the ARC, the NPY/Ag. RP neuron (green) produces NPY and Ag. RP while the POMC/CART neuron (red) produces POMC and CART.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
November 2017
Categories |
 RSS Feed
RSS Feed
